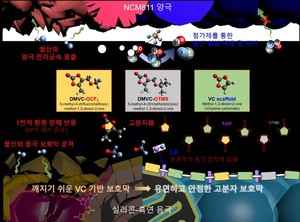
![The UNIST research team has developed an additive technology that can solve the problem of large-capacity battery life. A schematic diagram of the process of forming a cathode protective film and the effect of removing hydrofluoric acid by the developed additives (DMVC-OCF3, DMVC-OTMS).[사진= UNIST]](https://i0.wp.com/cdn.hellodd.com/news/photo/202102/91722_303104_4843.jpg?w=560&ssl=1)
Research that solved the life problem of large-capacity electric vehicle batteries with a small amount of additives is drawing attention. It maintains more than 80% of the initial capacity of the battery even after several hundred times of charging and discharging, and is evaluated as a performance surpassing existing additives.
UNIST (President Yong-Hoon Lee) announced on the 14th that the team of professors Nam-soon Choi and Sang-gyu Kwak of the Department of Energy and Chemical Engineering and Professor Sung-Yoo Hong of the Department of Chemistry have developed a battery electrolyte additive that can solve the instability of electrode materials, which has been considered a challenge in developing large-capacity lithium-ion batteries.
There is active research to replace the electrodes of commercial lithium-ion batteries with high-capacity materials such as silicon and high nickel and high nickel. However, the silicon negative electrode has a poor mechanical durability as the volume increases by more than three times during charging and discharging. High-nickel anodes also suffer from chemical instability.
The developed additive creates a protective film on the surface of the silicon mixed anode, which is flexible and stretchable like a rubber band, and has excellent lithium ion permeability (mobility). This is the reason why high-speed charging is possible by reducing mechanical overload caused by repeated volume changes of silicon. In addition, the additive removes hydrofluoric acid (HF) from the electrolyte to prevent the metal (nickel) inside the high nickel anode from leaking out. The amount of metal inside the anode determines the battery capacity.
The research team added the additive to a high-capacity battery composed of a high-nickel anode and a silicon mixed cathode. As a result, the initial capacity was maintained at 81.5% even after 400 charging and discharging. This is a 10-30% improved performance compared to the existing additives.
The co-first author Se-won Park, a PhD researcher in the Department of Energy and Chemical Engineering at UNIST, explained, “Even in the experiment of fast charging the battery within 20 minutes, only 1.9% capacity reduction was observed after 100 operations.”
Professor Nam-soon Choi said, “This achievement is the result of collaboration between designing and experimenting with material structures that can compensate for the disadvantages of existing additives (VC), simulations, and research on synthetic methods to actually make this material structure.” It suggested a new direction for development,” he pointed out the meaning of the study.
Professor Seongyu Hong explained, “The specific structure of the additive that forms the polymer protective film is a structure that is not well synthesized through general chemical reactions, so we solved the problem by going through an intermediate reaction.”
Professor Kwak Sang-gyu said, “The developed additive decomposes in the electrolyte to create an active ingredient (radical). This active ingredient reacts sequentially with other additive ingredients to create a flexible polymer protective film on the surface of the silicon electrode.” Professor Jaepil Cho of the Department of Energy and Chemical Engineering at UNIST participated in this research.
The research was conducted with the support of the Korea Institute of Energy Technology Evaluation and Planning’s energy technology development project and the Korea Research Foundation’s climate change response technology development project. The results were published in 5 characters in the international journal Nature Communications. It was also introduced as Editors’ Highlights selected by journal editors in recognition of their research excellence.
Send SNS article